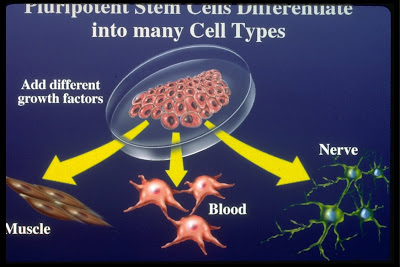
Finally the controversial stem cells were approved by the Food and Drugs Administration to be used to treat people whit spinal injuries, so this is how US doctors have begun the first official trial of using human embryonic stem cells, that have the potential to become in many of the different cell types found in the human body.
Geron, a biotech company located in the famous "Sillicon Valley" near San Francisco, is the company that get green light for these trials, they have spend $170m dollars on developing a stem cells (SC) treatment for spinal cord injuries. They start working whit human SC in 1999, when many predicted that some decades would be, before cell therapy would be approved for human clinical trials. Now that's a reality, but it will take some time to evaluate results, and more time and research to know if the therapy is safe and effective.
Every year 12,000 people suffer spinal cord injuries just in US, here in Mexico, through the TELETON, we know that a lot of people suffer this kind of injuries by causes like car accidents or falls.
To see more information : http://www.bbc.co.uk/news/health-11517680
It is very important to remember the bioethical implications in this kind of trials in human, remembering the unfortunate event were US doctors made human trials on Guatemalan population which included the infection of people whit sexually transmitted diseases.
Do you have any opinion???
10 comments:
hope to hear more views.
puessssss bueno, si la FDA ya lo aprobó y si los tratamientos serán en Estados Unidos (es decir, a cargo de doctores de verdad con gente de allá) no creo que sea comparable con lo de Guatemala. No sé, hasta me parece bueno, ya mucho se dice del beneficio de usar las células madre en muchos tratamientos. Creo que esta noticia es simplemente el siguiente paso.
salu2!!
Estoy de acuerdo contigo alejandra, No creo que este caso sea comparable con Guatemala, si bien, es cierto que hay que tomar en cuenta la cuestion etico que implica hacer los experimentos, este es un tipo de beneficio que dudo yo, que hubiera problemas serios comparados con ese acontecimiento. Ahora bien, la FDA ya lo aprobo y valdra la pena hacer las pruebas, con el consentimiento de las personas adultas. Este es un inicio necesario para terminar con el problema que aquja a un gran numero de personas que padecen estos problemas com lo menciona el articulo.
Ivette Salazar...
Las células madre han sido un caso algo polémico, pero en lo particular me parece que fue buena la idea de aprobación de las células madre, ya que pueden servir para curar muchas enfermedades que aquejan a la población, evitar el sufrimiento y al mismo tiempo tratar de mejorar la vida de las personas. Estoy de acuerdo con su utilización simpre y cuando se sigan normas específicas y claras. Incluso los gobiernos podrían ayudar teniendo bancos de células madre que sean donados de cordones umbinicales para que luego puedan ayudar a la población a combatir enfermedades.
me parece un mui buen y grande paso para la ciencia. Espero que sea el primero de muchos para este tipo de enfermedades, ya que son tantas personas que lo padecen, estoy en pro de ello ya que es para algo benefico, espero que si funcione seria un gran exito tanto para la medicina y una esperanza de vida mas para estas personas
No cabe duda que el uso de células madre es un gran avance científico pero el dilema ético que esto implica, es decir, la destrucción de embriones humanos, es muy complicado. La aprobación del uso de células madre puede llevar a conductas reprobables o moralmente dudosas y pueden surgir más dilemas. Por ejemplo: ¿Estarían de acuerdo en que las mujeres puedan vender sus embriones? ¿Y si lo hacen para beneficiar a algún ser querido? Además, hasta donde yo tengo entendido, cuando se ha experimentado el uso de células madre en animales, el cuerpo rechaza a estas células por ser genéticamente diferentes, y en la mayoría de los casos se produjeron tumores. Agradecería si alguien me podría aclarar si eso ya dejo de ser un problema. Saludos.
José Zavala
Me agrada escuchar esta noticia, siempre ha sido un tema difícil por la cuestión ética y por demás en lo religioso. Pero personalmente considero que ayudar a personas que padecen de estos problemas vale la pena, que sea através de este medio. La tecnología es punto y aparte de las creencias etc. Mientras se pueda ayudar el uso de celulas madre no es motivo de conflicto.
El conflicto surge en los posibles daños colaterales que pueden surgir de la par con los beneficios. Los ejemplos que se me vienen a la mente son la energía atómica y la dinamita. Claro que generaron grandes beneficios pero el costo no se puede minimizar. Creo que se debe ser más cauteloso en este terreno, porque la historia ha probado que a veces el hombre se apresura e incursiona en campos que no puede controlar.
Hola Luis, interesante lo que comentas, pero me parece que la obtención de las células madre de embriones no es planteada. Pero sería bueno investigar. Y sí, estoy de acuerdo, utilizar estos tratamientos puede traer opiniones contrarias de diversos grupos, ya las hay. Pero como bien dice José Zavala es punto y a parte. Interesante como el sentido del bien y el mal se modifica con el tiempo, hasta eso "evoluciona"... no les parece???
Salu2 a todos!!!
Según la entrada del blog están contemplando la utilización de células embriónicas, puesto que se menciona el dilema ético y la controversia que ha generado la consideración de su uso, además en el artículo del diario Reuters mencionan el uso de células de embriones con apenas algunos días de gestación, lo cual a mi parecer no puede llamarse asesinato aunque para muchas otras personas sea diferente y en varias religiones sea un crímen el uso de cigotos o mórulas creados in vitro. Pero bueno, debemos tomar en cuenta que hace años la fecundación in vitro también era considerada anti-ética.
El uso regulado de estas células simplemente disminuirá la manipulación ilegal y la experimentación con personas contra su voluntad como ha pasado tantas veces; y serguirá ocurriendo probablemente, sólo en menor cantidad. Todo esto abre para mi todo un nuevo panorama de abusos y mercado negro, jamás ha existido desarrollo sin daño. La ciencia todavía tiene un largo recorrido y solo la experimentación puede proveer de nuevo conocimiento, ahora hay que esperar que repercusiones puede traer; los beneficios, de funcionar óptimamente, pueden ser enormes, aun mayores a los efectos negativos.
Post a Comment